

Grasshoppers are notoriously difficult to preserve. When air-dried, their soft bodies often shrink, collapse, or darken, producing poor-quality specimens.
This page describes my method for pinning grasshoppers and preserving them using a simple freeze-drying technique developed for a standard chest freezer.
Orthoptera are more difficult to preserve than many insects because their bodies contain a large amount of soft tissue. When specimens dry at room temperature, water leaves the body unevenly. As a result, abdomens often collapse and shrivel, and vibrant colors are lost, sometimes darkening or turning black as decay begins.¹
Preserving specimens in alcohol avoids collapse but introduces other problems. Many pigments fade in liquid preservatives,² and fine structures such as setae or ocelli become difficult to see because the refractive index of the surrounding liquid reduces visual contrast.³

Comparison of air-dried (left) and freeze-dried (right) specimens of Dactylotum bicolor and Leprus wheeleri.
Freeze-drying removes water while the specimen remains frozen. Ice within the insect turns directly into water vapor through a process called sublimation. Because the tissues remain frozen during drying, their structure stays in place as moisture is removed.
Studies have shown that freeze-dried insects retain their natural shape, surface detail, and coloration much better than specimens that are air-dried. Researchers have noted the remarkably lifelike appearance of freeze-dried specimens.² ³ ⁴ ⁵ ⁶
Green rarely preserves well long-term. Chlorophyll, the compound responsible for green pigmentation, gradually breaks down during specimen storage.⁴
Freeze-drying removes water from a frozen specimen through sublimation, a phase change in which ice turns directly into water vapor without first melting into liquid water.
For sublimation to occur, the vapor pressure of ice must be higher than the partial pressure of vapor in the surrounding air. When this condition is met, water molecules escape from the ice and enter the air as vapor.
Temperature — Warmer sub-freezing temperatures increase the vapor pressure of ice, which speeds up sublimation.
Dry air — Keeping the surrounding air dry lowers the partial pressure of water vapor, allowing moisture to leave the specimen more efficiently.
Low pressure — Lower pressures, such as in a vacuum, reduce the number of air molecules (e.g., oxygen and nitrogen). This allows escaping water vapor molecules to travel more freely without colliding with other gas molecules.
Freeze-drying is normally performed using specialized equipment that carefully controls temperature and pressure while maintaining a strong vacuum. These machines accelerate sublimation and allow specimens to dry rapidly. However, commercial freeze-dryers are expensive and out of reach for most hobbyists and small collections.
Fortunately, freeze-drying can still occur in a household freezer if specimens remain frozen in a sufficiently dry environment. Ice within the specimen can gradually sublimate over time as long as water vapor is continually removed from the surrounding air.
To test whether this process could be accelerated, I conducted an experiment using a chest freezer and several modern tools designed to control temperature and remove moisture.
Equipment Used:

Two standard 7.0 cu. ft. chest freezers were used for the experiment.

Temperature controller used to maintain a warmer sub-freezing temperature.

The experiment tested activated alumina, silica gel, and molecular sieve.

Zwilling Fresh & Save glass containers were used to reduce air pressure around the specimens.
Nine female Trimerotropis pallidipennis specimens were used in the experiment. They were killed by freezing and then allowed to thaw until pliable. Each specimen was then pinned and positioned on a piece of foam. Each specimen was weighed, subtracting the weight of the foam and pin.
Individual pinned specimens were placed inside containers along with a desiccant. The containers were then placed inside the chest freezers.
Each week the specimens were weighed to measure moisture loss. After 16 weeks, the specimens were removed from the freezer and allowed to dry in desiccant containers at room temperature for two additional weeks in order to determine their fully desiccated weight.
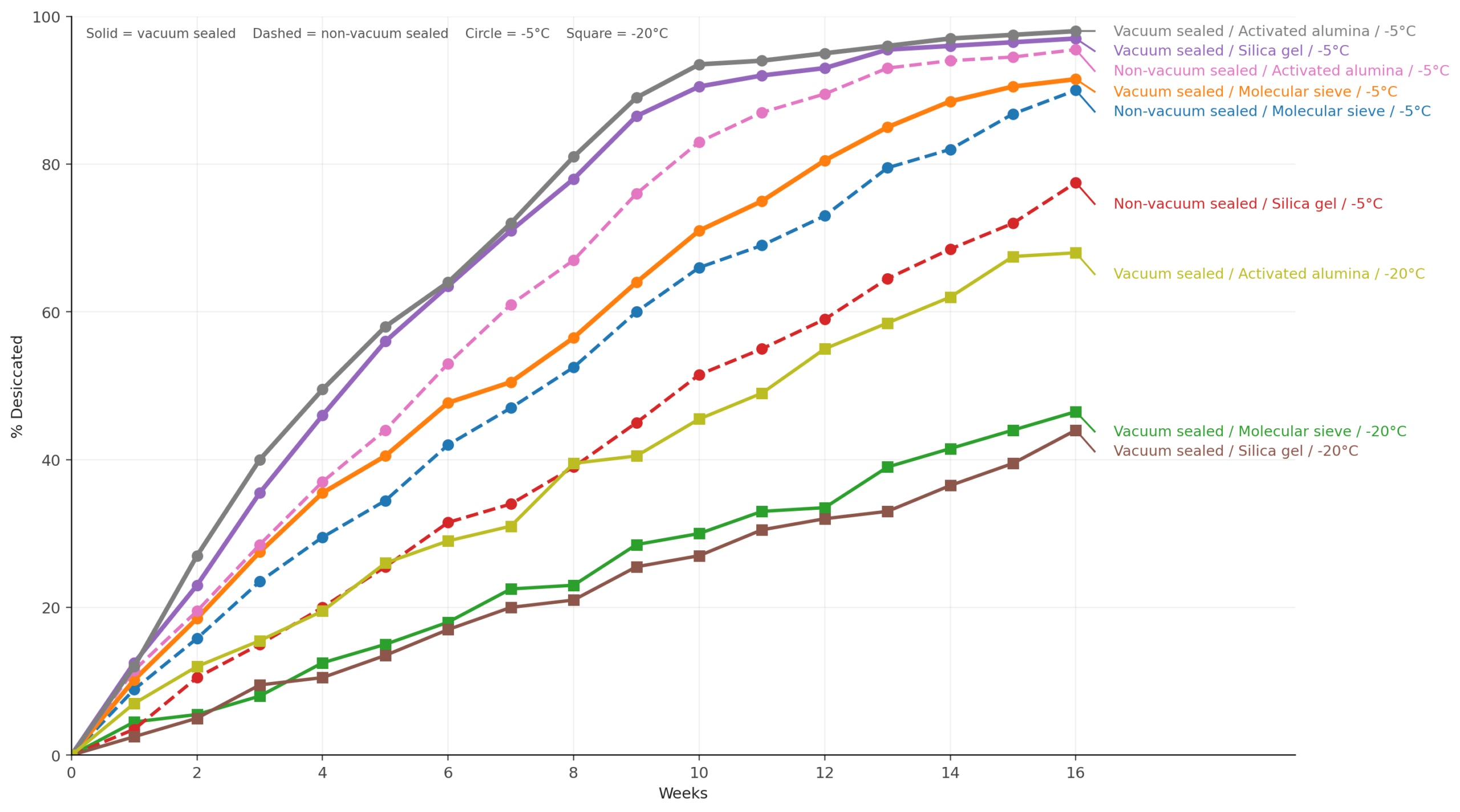
Figure 1. Moisture loss of pinned female T. pallidipennis grasshopper specimens during freeze-drying. Lines show the percentage of total desiccation achieved under different experimental conditions.
Solid lines represent vacuum-sealed containers, while dashed lines represent non-vacuum containers. Circles indicate −5 °C freezer conditions, and squares indicate −20 °C.
The fastest freeze-drying occurred at −5 °C using activated alumina in a vacuum-sealed container, reaching over 90% desiccation in approximately 10 weeks.
According to Flaschka and Floyd, it may be sufficient to freeze-dry specimens to roughly 50% moisture loss and then complete desiccation outside the freezer at room temperature.⁴
Roe and Clifford noted that drying time varies between species and is not necessarily related to specimen size.²
In my own unpublished experiments, males typically desiccate about one month sooner than females. As a general rule, I leave males in the freezer for about two months and females for roughly three months.
Freeze-dried insects can still be relaxed and repositioned if necessary.
Based on the results of the experiment, I now freeze-dry my specimens at −5 °C using activated alumina.
However, I do not use vacuum-sealed containers. In practice, it is more convenient to use a large airtight container that can hold many specimens at once. I use Araven 03036 4″ deep full-size containers with airtight lids, which fit perfectly inside the 7.0 cu. ft. chest freezer.
To pin the grasshoppers, I made custom foam pads that use magnetic sheets to hold the wings in place.
The images below show additional details of the setup and resulting specimens.
This simple freeze-drying method allows high-quality grasshopper specimens to be prepared using inexpensive equipment available to most collectors.
¹ Meryman, H. T. (1960). The physics of freeze-drying.
² Roe, R., & Clifford, C. (1976). Color preservation and morphology in freeze-dried insects.
³ Woodring, J., & Blum, M. (1963). Freeze-drying arthropods for taxonomic study.
⁴ Flaschka, M., & Floyd, R. (1969). Freeze-drying insects in a household freezer using silica gel.
⁵ Dominick, R. (1972). Preservation of insects by freeze-drying.
⁶ Albrecht, M. (1994). Freeze-drying aphids for preservation of morphology.
Author: Adam Retzer
Date: April 2026